IGNOU BCHCT-131- Solved Assignment
Are you looking to download a PDF soft copy of the Solved Assignment BCHCT 131 Atomic Structure, Bonding, General Organic Chemistry is the right place for you. This particular Assignment references the syllabus chosen for the subject of Hindi, English, for the Jul 25 – Jan 26, Jul 24 – Jan 25 session. The code for the assignment is BCHCT-131 and it is often used by students who are enrolled in the BSC (Honours), IGNOU CBCS Solved Assignments Degree. Once students have paid for the Assignment, they can Instantly Download to their PC, Laptop or Mobile Devices in soft copy as a PDF format. After studying the contents of this Assignment, students will have a better grasp of the subject and will be able to prepare for their upcoming tests.
PLEASE MATCH YOUR ASSIGNMENT QUESTIONS ACCORDING TO YOUR SESSION
IGNOU BCHCT-131 (January 2026 – December 2026) Assignment Questions
1. Explain Geiger and Marsden’s a-particle experiment using a suitable diagram
2. (a) Name different lines exhibited in the atomic spectrum of hydrogen.
(b) Write the de-Brogile relationship for dual nature of matter. Also define matter waves.
3. Briefly discuss photoelectric effect. Also give the explanation of this effect given by Einstein.
4. (a) List the applications of Schrodinger equation.
5. What are four quantum numbers? Give their significance.
6. (a) Explain the stability of half-filled and completely filled orbitals giving suitable examples.
(b) Give any two examples of atoms showing anomalous electronic configurations.
7. State and illustrate Fajan’s rules giving suitable examples.
8. (a) What is electronegativity? Arrange the following in the decreasing order of their electronegativities: K, F and I
(b) Compare the characteristics of ionic and covalent compounds.
9. (a) Write the resonance structures of dimethylsulphone.
(b) Give the type of hybridisation in the following molecules:
10 (a) Draw py-py combination of atomic orbitals to give the suitable molecular orbitals.
(b) Calculate the bond order of F2 molecule from its molecular orbital configuration.
PART-(B)
(b) How will you distinguish maleic acid and fumaric acid?
(b) Give example of a meso compound. Also comment on its optical activity giving reason.
13. Draw the energy profile for various confirmations exhibited by cyclohexane. Also explain the relative energies of various conformations.
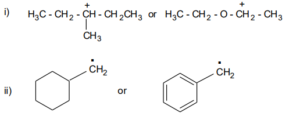
14. Explain the relative stability of the following carbocations:
(i) methyl carbocation
(ii) a tertiary carbocation
(iii) a primary carbocation
(iv) a secondary carbocation
15. (a) Differentiate between the basicity and nucleophilicity giving suitable examples
(b) Briefly explain the mechanism of β-elimination,
16. (a) Briefly explain Kolbe’s electrolytic method for the synthesis of alkanes.
(b) What is aromatisation of gasoline? Illustrate giving appropriate reactions.
17. State and explain Saytzeff rule giving a suitable example.
18. Explain retro Diels-Alder reaction giving a suitable example.
19. (a) (i) State Markowinkoff’s Rule.
(ii) What is peroxide effect? Explain.
(b) Write the reactions for the hydroboration of propene followed by the oxidation by alkaline H2O2.
20. (a) How will you prepare 3-octyne from 1-hexyne? Write the steps involved.
(b) Write the products of oxidation of 2-hexyne using alkaline KMnO4.
IGNOU BCHCT-131 (January 2025 – December 2025) Assignment Questions
IGNOU BCHCT-131 ASSIGNMENTS DETAILS
| University | : | IGNOU (Indira Gandhi National Open University) | |
| Title | : | Atomic Structure, Bonding, General Organic Chemistry and Aliphatic Hydrocarbons | |
| Language(s) | : | English, Hindi | |
| Code | : | BCHCT-131 | |
| Degree | : | BSC (Honours), BSCG, BSCM | |
| Subject | : | Chemistry | |
| Course | : | Core Courses (CC) | |
| Author | : | ignouedumart.com Panel | |
| Publisher | : | Distance Gyan Publishing House Pvt. Ltd. |
Call us: +91 9466323363





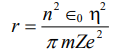





Reviews
There are no reviews yet.